- 0086-21-56469616
- 0086-18019205509
- minstar@minstargroup.com
- Language:English
- English
Your Location:Home >Products >Fine Chemicals >59-05-2

Appearance:powder
Throughput:1|Metric Ton|Day
pd_productuse:intermediate
Delivery Time:in stock
Purity:99%
Methotrexate Basic information
Treatment of cancer and rheumatoid arthritis History of discovery Immunosuppressants Pharmacological effects Pharmacokinetics Clinical application Dosage Adverse reactions and precautions Drug Interactions Chemical Properties Production method
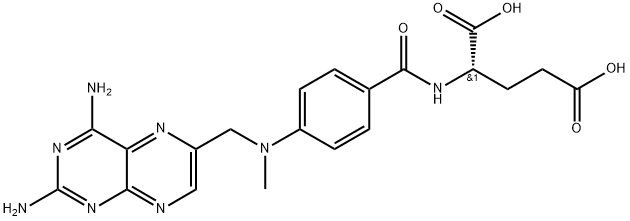
Product Name: Methotrexate
Synonyms: METHOTREXATEFORINJECTION;sodium 4-[[4-[(2,4-diaminopteridin-6-yl)methyl-methyl-amino]benzoyl]am ino]-5-hydroxy-5-oxo-pentanoate;METHOTREXATE(N-(4-(((2,4-DIAMINO-6-PTERIDINYL)METHYLAMINO)BENZOYL)-L-GLUTAMICACID);4-Amino-N10-methylfolic acid;Glutamic acid, N-[p-[[(2,4-diamino-6-pteridinyl)methyl]methylamino]benzoyl]-, L-(+)- (8CI);L-Glutamic acid, N-[4-[[(2,4-diamino-6-pteridinyl)methyl]methylamino]benzoyl]- (9CI);L-Methotrexate;Methotrexat-Ebewe
CAS: 59-05-2
MF: C20H22N8O5
MW: 454.45
EINECS: 200-413-8
Mol File: 59-05-2.mol
Safety Information
Hazard Codes T,F
Risk Statements 61-25-36/38-46-39/23/24/25-23/24/25-11
Safety Statements 53-26-36/37-45-36/37/39-36-16
RIDADR UN 2811 6.1/PG 3
WGK Germany 3
RTECS MA1225000
F 3-8-10
TSCA Yes
HazardClass 6.1(b)
PackingGroup III
HS Code 29335995
Hazardous Substances Data 59-05-2(Hazardous Substances Data)
Toxicity LD50 oral in rat: 135mg/kg
CAS:885523-08-0
CAS:517920-73-9
CAS:69-53-4
CAS:6180-61-6